With 20+ years of pathology expertise and regulatory insight, we help you navigate the pitfalls that often derail medical device development: incomplete planning, inadequate sample collection or processing, need for advanced techniques and specialized stains that compromises data quality, and the costly assumption that all suppliers offer equivalent pathology services.
We have conducted more than 1,000 Non-GLP and GLP medical device pathology studies across multiple therapeutic areas, delivering the specialized expertise and proactive guidance to move programs forward from day one.
Choosing the right CRO means finding a partner who treats your project as their own priority. StageBio works as an extension of your team, combining consultative expertise with transparent collaboration to accelerate your path from study to submission. Our pathologists and scientists provide strategic insights beyond just data generation and reporting, helping guide the best approach from protocol design through regulatory filing. With decades of experience and a commitment to open dialogue and clear timelines, we deliver submission-ready data and reports aimed to contextualize the data advance your program with confidence.
Cardiovascular
Device Studies
Nervous System
Device Studies
Musculoskeletal & Dental Device Studies
Skin & Wound Healing
Device Studies
bringing breadth and depth, accelerating turnaround times and strengthening regulatory confidence
delivering validated insights, regulatory-grade interpretation, and credibility you need to drive programs forward
We offer a range of specialized GLP-compliant pathology services for medical devices to support your project from preclinical studies through regulatory submission. With our proven experience and deep expertise, we deliver the quality, timelines, and regulatory assurance you need, while providing strategic insights that add true value to your program.
Download our flyer for a full list of services we offer for medical device evaluation.

We conduct robust non-clinical evaluations (necropsy, histopathology, morphometry, image analysis) of devices and biomaterials in any tissue but specially in the following areas:
We offer a wide platform including bespoke histopathology techniques, imaging, and analytics for:
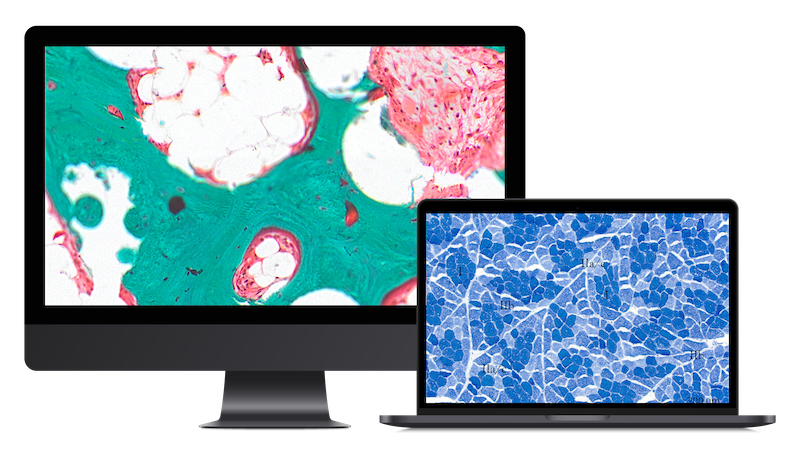
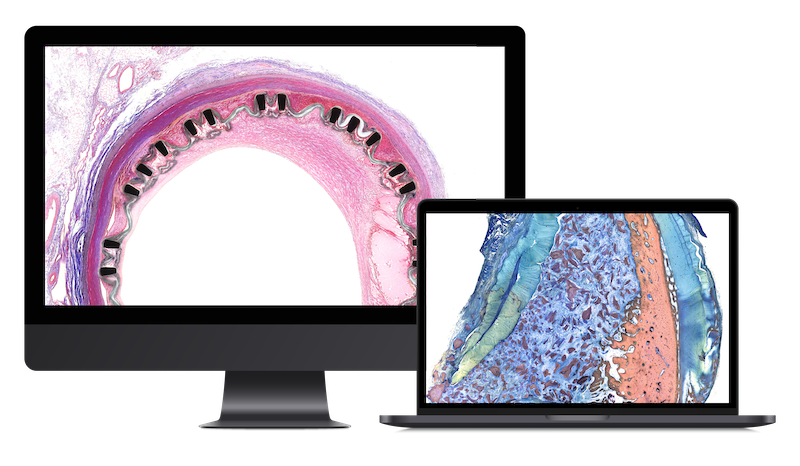
Our quality-driven scientific team is dedicated to open collaboration and flexible support. StageBio provides a tailored approach with clear, insightful interpretation to support you with:
Meet with our pathology team to discuss how StageBio's expert support can keep your study on track and deliver the quality data you need, when you need it.
Trusted Histopathology Support for Medical Device Development
Key Pathology Considerations for Successful Preclinical Medical Device Studies, from Protocol to Report
Histopathological Evaluation of Orthopedic Medical Devices: An Overview of Tissue Collection and Sample Storage
Medical Device Pathology: The 7 Common Mistakes We Help You Avoid
“I have worked with a number of pathologists now and I really appreciate the systematic approach to grading, the recognition that one event in one nerve bundle in one slide does not constitute a generalizable phenomenon.” Director of Pathology, Contract Research Organization
“There is literally no other group with this degree of knowledge, ability to put data in a context, and to be honest and direct with their client about the data. This really sets [StageBio] apart from other groups.” Study Director, Medical Device Organization
“At StageBio, what makes us stand out is the way we partner closely with our clients, supporting them with their study design input, specialized laboratory methods, bespoke morphometric evaluations, and comprehensive pathology reports. Our sponsors depend on us for precision, quality, and GLP compliance to ensure their programs move forward with confidence.” Jaime Paulin, DVM, MS, DACVP, StageBio, Senior Pathologist