Learn how integrating toxicologic pathology best practices into medical device evaluation can improve biological assessment, strengthen regulatory outcomes, and reduce study risk.
Medical device innovation moves fast, and nonclinical safety evaluation is struggling to keep up. As therapy modalities grow more complex, the rigid, engineering-centric frameworks currently used to assess medical device biocompatibility are falling short of capturing real biological risk.
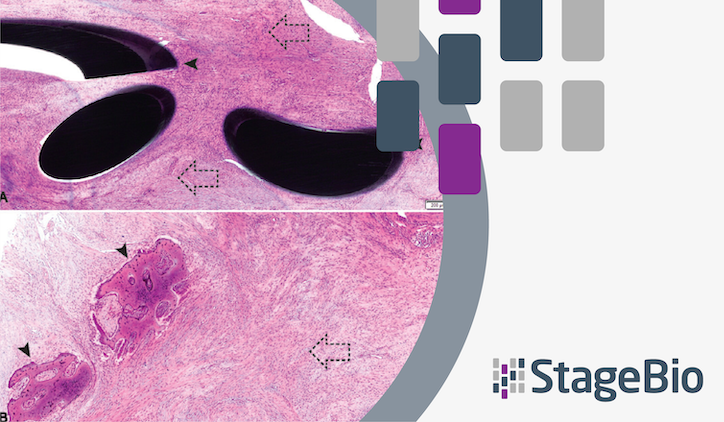
In a recent publication by Funk et al., StageBio pathologist Dr. Jaime Paulin, ACVP, and consulting pathologist Dr. Abraham Nyska are among the co-authors making the case for a better approach. Published in the November 2025 edition of Advanced Healthcare Materials, the paper argues that early involvement of experienced toxicologic pathologists, flexible study designs, and biologically informed scoring rubrics produce more accurate, regulatory-ready safety data than the one-size-fits-all methods currently in wide use.
Key topics covered in the paper include the limitations of ISO 10993 biocompatibility scoring versus adversity-based assessment, the risks of applying rigid rubrics from ISO 10993-6 (Annex E), and practical recommendations for improving pathology evaluation in nonclinical medical device studies.
Evaluating medical devices using toxicologic best practices to capture biological response more accurately
For biotech, pharma, and device developers navigating complex regulatory pathways, engaging experienced toxicologic pathologists early in study design is one of the most impactful steps you can take. Moving away from rigid checkbox compliance toward flexible, biologically relevant best practices means stronger data for regulatory submissions, and fewer costly surprises down the line.
Ultimately, harmonizing medical device evaluation with established toxicologic pathology principles is a win across the board: better risk assessment, reduced unnecessary animal testing, and safer outcomes for patients.
Access the full review published in Advanced Healthcare Materials
Download the review paper here.
Work with StageBio's Medical Device Pathology Experts
Looking to improve the pathological evaluation of your next medical device study? Our team of experts is ready to help you design rigorous, regulatory-ready studies from the ground up.