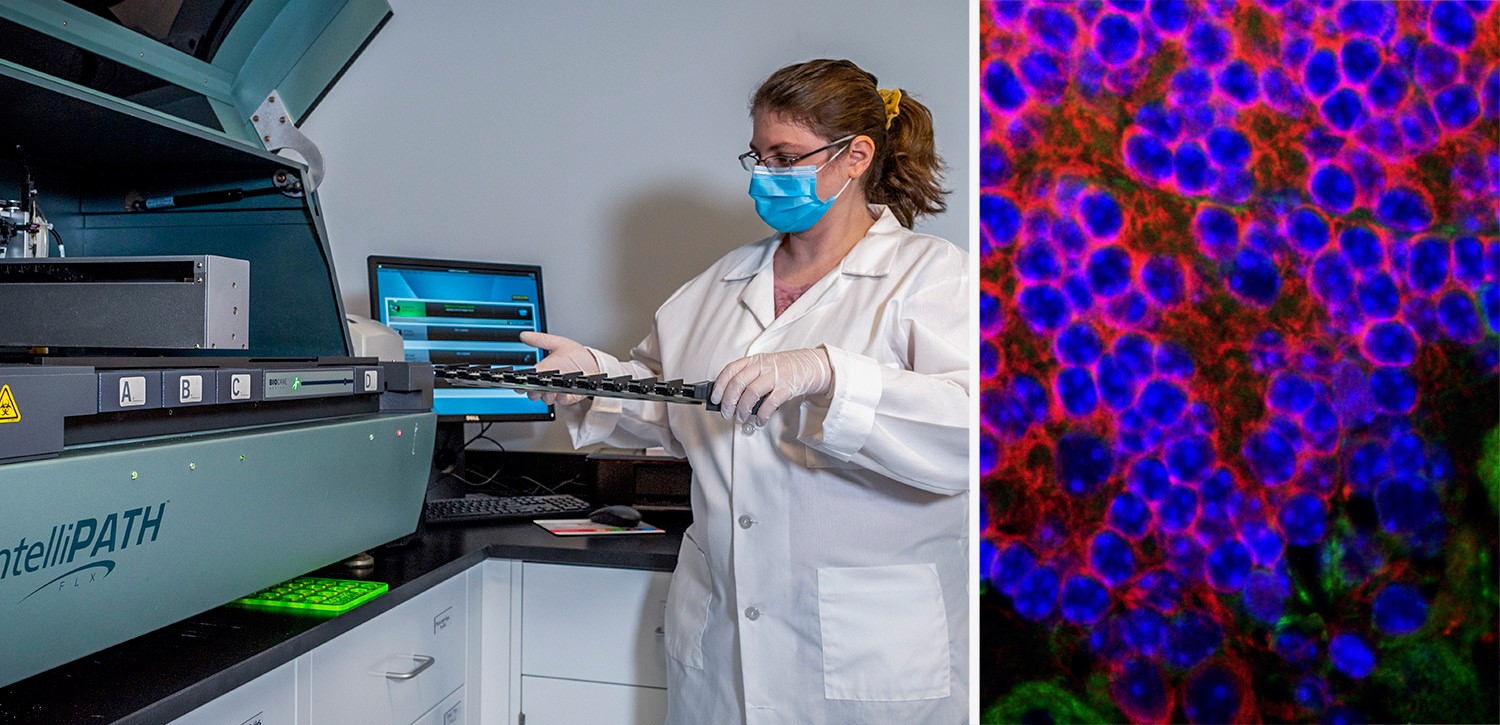
StageBio Announces Opening of New Facility in Greater Boston Area
State-of-the-art facility in Marlborough, MA brings full range of
preclinical and translational histopathology services close to biotech hub.
MARLBOROUGH, MA, August 12, 2020—StageBio, a leading provider of GLP-compliant necropsy, histology, pathology, and specimen archiving services for both preclinical and clinical research, has opened a new facility close to the biotechnology and medical research centers of Cambridge and Boston. This new facility will offer local access to the full range of services, including molecular and investigative pathology, multiplexing IHC, whole slide scanning, and computerized histomorphometry to support academic, pharmaceutical, and medical device development.
The nearly 10,000-square-foot facility brings industry-leading histopathology expertise closer
to the Boston/Cambridge area, expanding StageBio’s local offerings to include a robust menu
of special stains, immunohistochemistry, immunofluorescence, in situ hybridization, and multiplexing techniques, with the option to deploy assays across a variety of instruments and reagents. Courier services for the greater Boston/Cambridge area to the new facility are available to ensure specimen integrity and accelerate turnaround times for tissue specimens, slides, and histopathology reports.
The GLP-compliant facility features a state-of-the-art laboratory staffed by consultative subject matter experts spanning the preclinical, translational, and clinical continuum, led by Director of Investigative Pathology Elizabeth Hutto, DVM, Ph.D., DACVP. A dedicated study management team assigns skilled technicians, imaging specialists, and pathologists to each project. Guiding studies from protocol development through necropsy, histology, pathology, whole slide scanning, and image analysis, StageBio delivers high-quality detailed information and reports required to advance research programs.
“The Boston/Cambridge area represents the industry's richest wellspring of drug discovery
and innovation”, said StageBio CEO Tom Galati. “We believe the opportunity to tap into
best-in-class talent and network with a variety of pharmaceutical and biotech partners positions StageBio as a preferred provider of both specialized and general histopathology services.”
Click here for a pdf of the press release.
About StageBio
StageBio is a leading provider of GLP-compliant necropsy, histology, pathology, and specimen archiving services for the biopharmaceutical, medical device, academic, and contract research industries. The company operates six GLP laboratories as well as two GLP specimen archiving facilities in the U.S., with substantial continued investment in facility and technology infrastructure to meet the growing demand for high-quality histopathology services globally. StageBio has a team of 25+ board-certified veterinary pathologists and more than 50 laboratory technicians on staff supporting our unified commitment to quality, scientific integrity and client satisfaction.
Learn more at stagebio.com