The International Harmonization of Nomenclature and Diagnostic Criteria for Lesions Project (INHAND) has published its overview of nonproliferative and proliferative lesions of the rabbit in the Journal of Toxicologic Pathology. StageBio's senior pathologist, Dr Brett Saladino, is a contributing author to this publication.
It provides a standardized international nomenclature for classifying microscopic lesions observed in most tissues and organs from laboratory rabbits used in nonclinical safety studies. These terms, as well as diagnostic criteria, found in the publication are applicable for the most commonly used strains of laboratory rabbits: the New Zealand White (non-pigmented), the New Zealand Red (pigmented), and the Dutch Belted (pigmented) rabbits.
The publication focuses on providing nomenclature and diagnostic criteria, arranged by body system in alignment with the INHAND publications for rodent pathology, with additional descriptions of gross and microscopic anatomy of rabbits. Findings and lesions that are unique to rabbits are described in greater detail. The publication also includes charts showing general incidence of lesions and 56 full color illustrations.
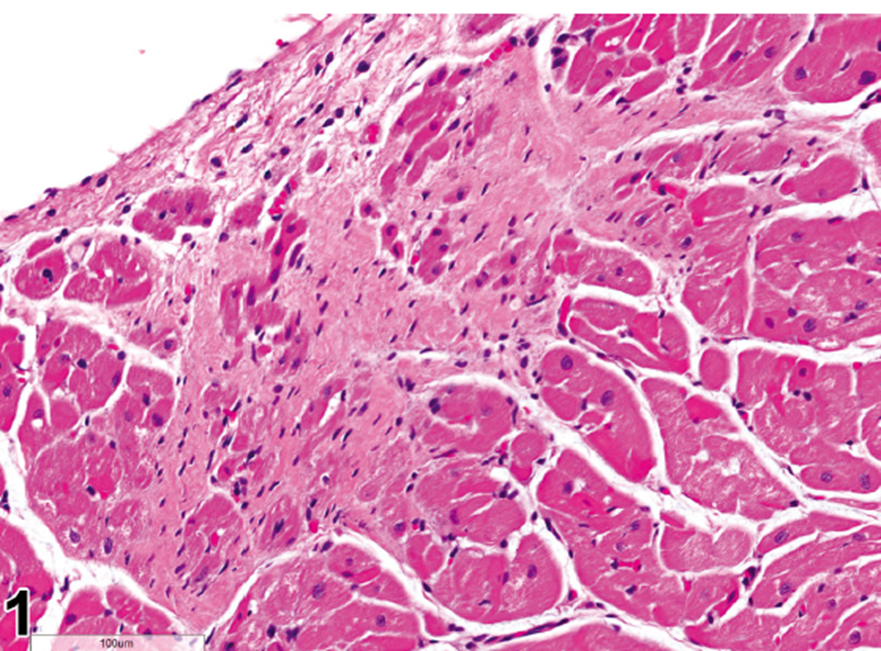
**Page 273**
Publication authors:
- Alys E Bradley
- Lyn Miller Wancket
- Matthias Rinke
- Margarita M Gruebbel
- Brett H. Saladino
- Kenneth Schafer
- Osamu Katsuta
- Begonya Garcia
- Franck Chanut
- Katherine Hughes
- Keith Nelson
- Lauren Himmel
- Elizabeth McInnes
- Adrienne Schucker
- Kazuyuki Uchida
Download
Contact us and we will update you when the full publication is ready: INHAND Nonproliferative and Proliferative Lesions of the Rabbit, at info@stagebio.com.